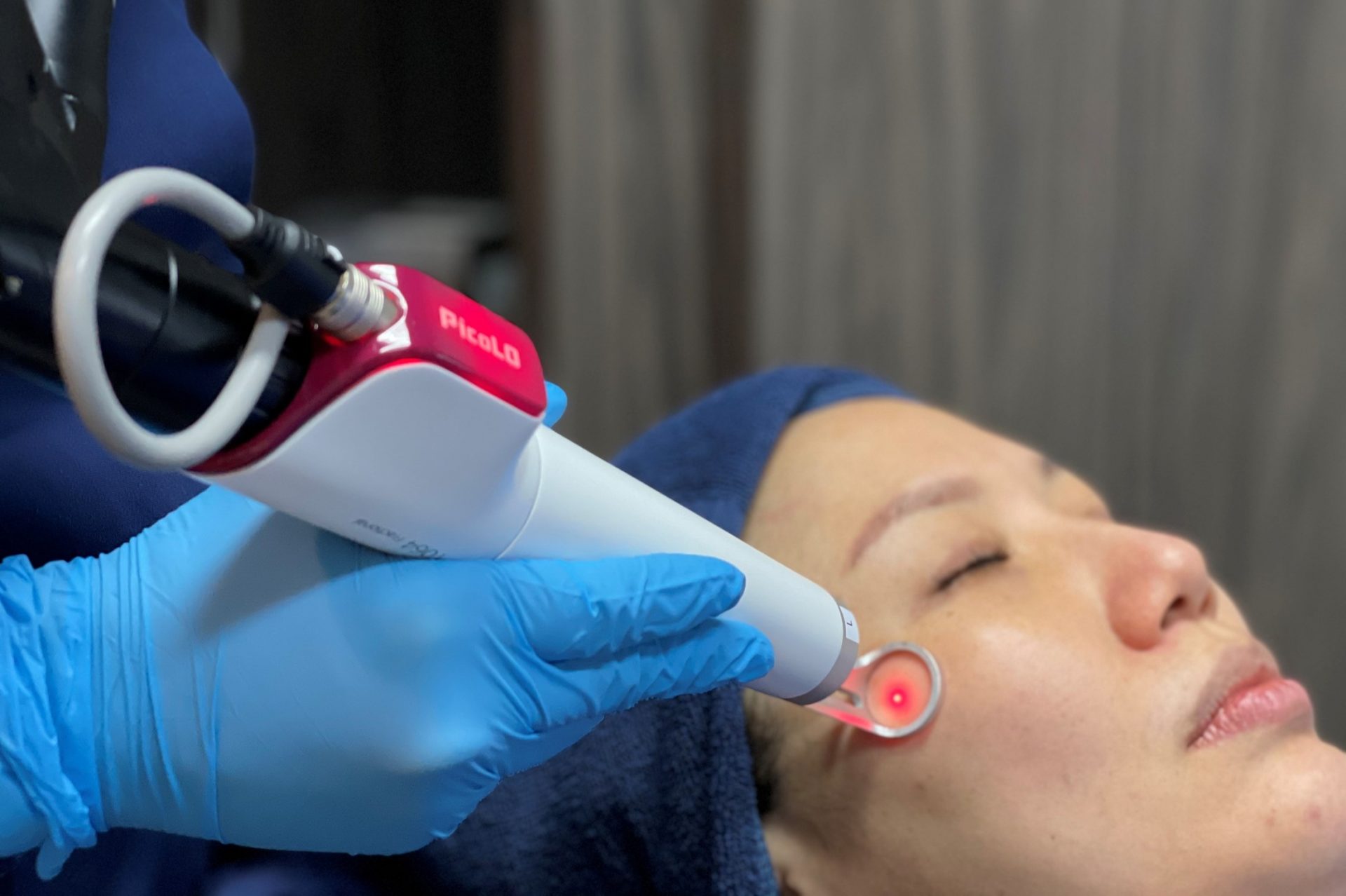
LASEROPTEK Co., Ltd. Receives FDA 510(k) Clearance for its Next Generation PicoLO Premium™ Picosecond Nd:YAG Laser – LASEROPTEK

LASEROPTEK - The PicoLO has recently received premarket clearance by the U.S. Food and Drug Administration (FDA) on demonstrating its capabilities in treating benign pigmented lesions, acne scars, and wrinkles both safely

LASEROPTEK - LASEROPTEK's PicoLO™ picosecond pulse Nd:YAG laser has been featured in Springer Nature's latest Updates in Clinical Dermatology textbook series' "Acne: Current Concepts and Management". Of the 17 studies conducted on